


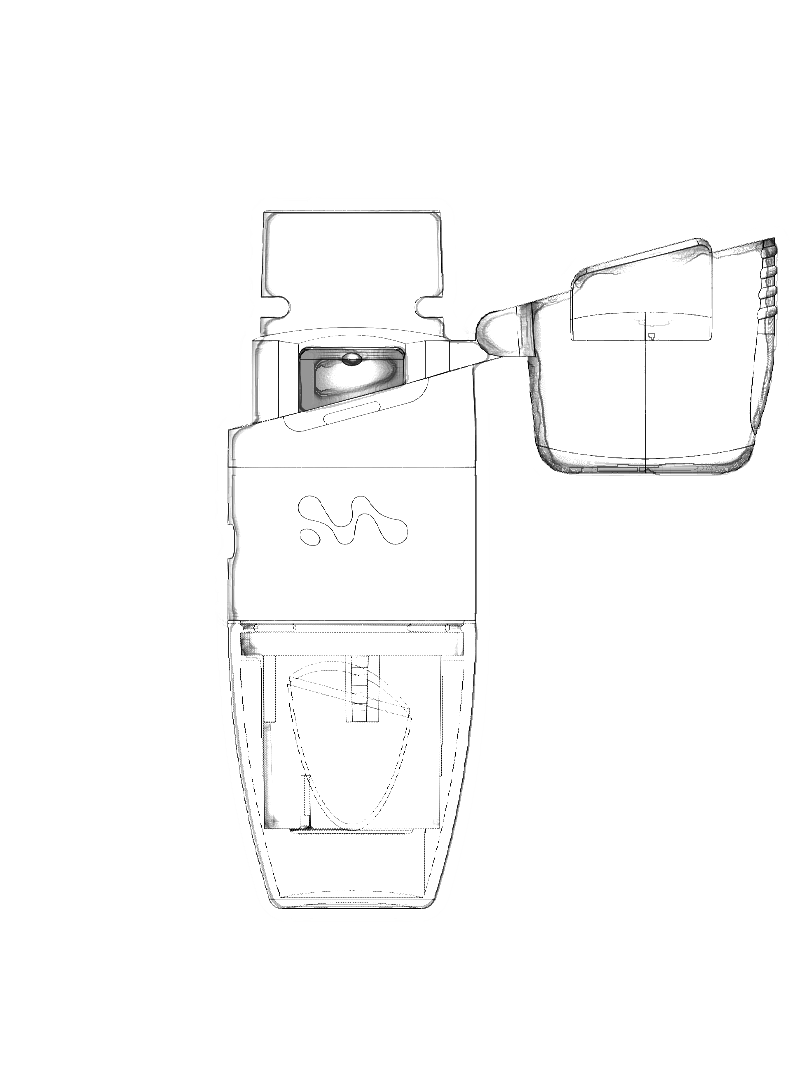
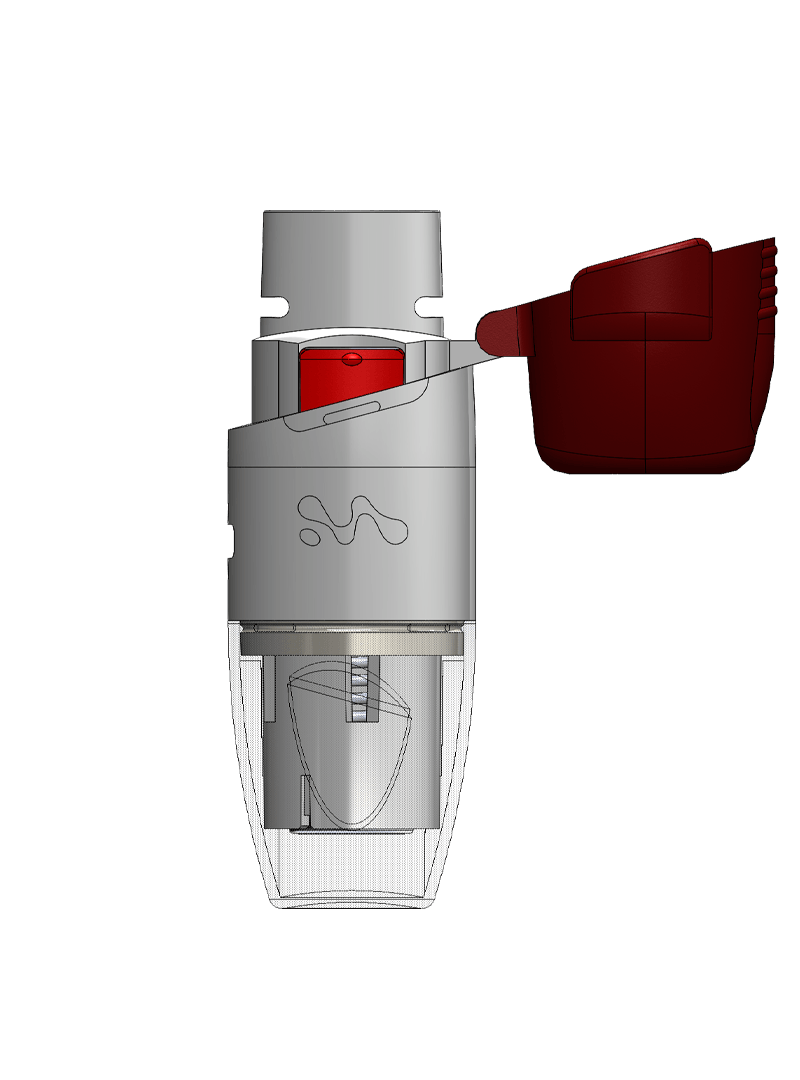

Merxin Ltd designs and supplies generic and customised inhaler devices. We redefine Inhaler Technology: multidose dry powder inhalers, capsule dry powder inhalers, soft mist inhalers, devices tailored to deliver cannabinoids to the lungs. We are the OINDP Experts.
Our customers take our devices and combine them with their drug formulation to make final dosage forms that are supplied to users and patients. We create, develop and supply inhalers for evaluation through to commercial supply.
EXPLORE OUR EXPERTISE“We are not a cost centre. We help you generate revenues”
Flexibility with the supply chain enables Merxin Ltd to make use of the best manufacturing opportunities in the world.
“Our expertise saves customers money and time to develop, file and market inhaled dosage forms”
The Merxin Ltd team has 100+ years of experience in OINDPs and many filings: we will share our knowledge with you
“We will create the records you need to file your finished dosage forms across the world”
Our ISO13485:2016 certification is your passport to quality assurance.
“We take problems away from our customer. We are part of the solution”
Proven device designs teamed with Merxin Ltd manufacturing experience reduce risk and the time to market.
The pharmaceuticals market is Merxin Ltd's native land. The team at Merxin Ltd has been working in and for the pharmaceutical industry for decades, designing devices, formulations, planning clinical studies and filings for the treatment of Asthma, COPD, IPAH, cystic fibrosis etc...
LEARN MOREOur body would not function were it not for its endocannabinoid system. Cannabis and its derivatives are now being brought back to the natural pharmacopeia...
LEARN MOREOur devices save customers money and time to develop, file and market inhaled dosage forms. We take problems away from our customer. We do it so you don’t have to. We are a trusted partner. We are part of the solution.
EXPLORE OUR PRODUCTS